- About us
- Organisation
- The Helsinki Convention
- Contracting Parties
- Chairmanship
- Secretariat / Staff
- Observers
- Internal rules
- Partners
- Vacancies
- Contact us
- HELCOM at work
- Groups
- Projects
- Ministerial meetings
- Events
- HELCOM Stakeholder Conferences
- HELCOM Stakeholder Conference 2025
- Baltic Stakeholder Conference 2024 – PROTECT BALTIC
- Baltic Stakeholder Conference 2023
- Baltic Stakeholder Conference 2022 – Climate Change in the Baltic Sea
- HELCOM Stakeholder Conference 2021
- HELCOM Stakeholder Conference 2020
- Marine Litter Stakeholder Conference 2017
- HELCOM Stakeholders Conferences (2006 – 2010)
- Events 2025
- Events 2024
- Events 2023
- Events 2022
- Events 2021
- Events 2020
- Events 2019
- Events 2018
- Events 2017
- Events 2016
- Events 2015
- Events 2014
- Events 2013
- HELCOM 50 years
- HELCOM 40 years
- HELCOM Stakeholder Conferences
- News & Media
- Publications
- Recommendations
- Home
- Baltic Sea knowledge
- Holistic Assessments (HOLAS)
- Biodiversity
- Eutrophication
- Hazardous Substances
- Maritime
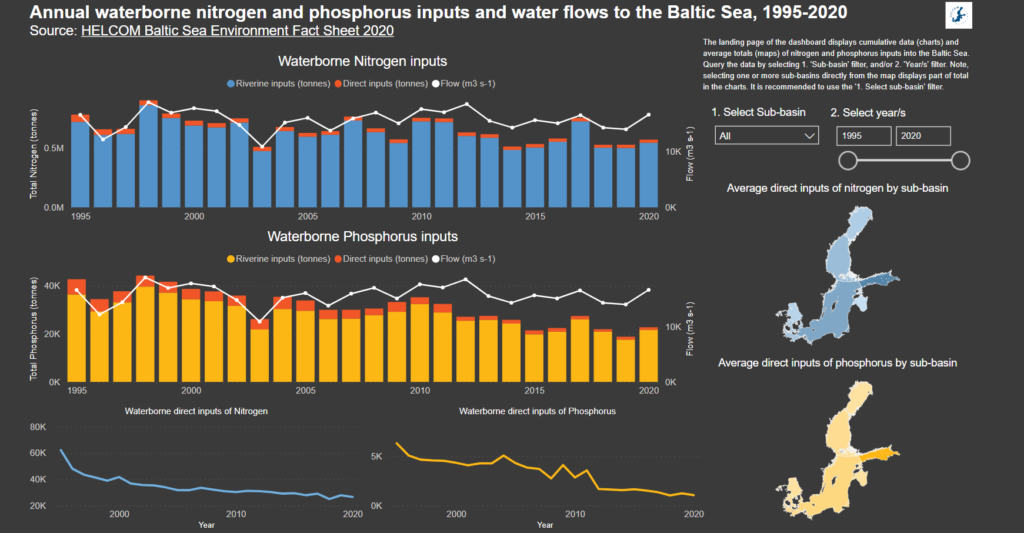
- Environment Fact Sheets
- Data
- Pollution Load Compilations
- Indicators
- Action areas
- Agriculture
- Fisheries
- Industrial & Municipal Releases
- Marine Litter & Underwater Noise
- Marine Protected Areas
- Maritime Spatial Planning
- Monitoring and Assessment
- Response to Spills
- Species and Biotopes
- Shipping
- Baltic Sea Action Plan
- Baltic Rescue – Match & Track
- Ecosfera Baltica
- Ecosfera Baltica rulebook
Baltic Marine Environment
Protection Commission